- Blog
- Toshiba r830 bluetooth
- Dirt 4 reviews
- Free download hunting unlimited 2014
- Coins generator fifa 16 no survey
- Vsco image id
- Planet coaster requirements
- Pinnacle studio 15 versione prova
- Use hec ras
- Pokemon sun moon logo
- Microsoft toolkit 2-6-7 telecharger
- Tom jerry cartoon in urdu
- Apps like camerafi live
- Download muv luv steam 18- patch
- 1- imperialism 2- colony 3- social darwinism 4- civilize 5- superior 6- barbaric 7- empire
- Netgear n150 wireless usb adapter wna1100 version 1-5
- Persona 5 pc full free download mega
- Attack on titan anime soundtrack
- Ralink rt2870 mac driver
- I don t want to be a player no more reggae
- This way that mario party 2
- Carbon 14 half life
- Matrix path of neo pc free download
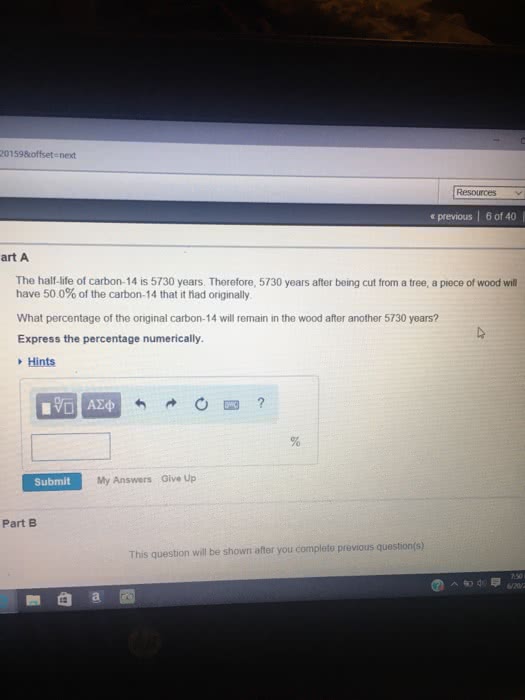
#CARBON 14 HALF LIFE HOW TO#
In the coming section, let us understand the different types of reaction, and how to derive its half-life reaction. Half-life varies between different types of reactions. Same way, time can be determined with the knowledge of concentration and rate constant.

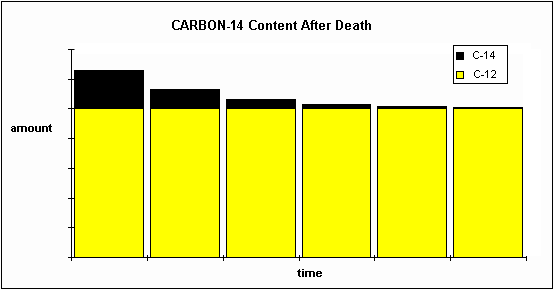
In pharmacokinetics, this principle of a half-life has a key role in the drug administration tests into the target, especially in the phase of elimination, where half-life is used in the calculation of time as to how fast a drug volume decrease in the given target after the reactant has been absorbed. Half-life principle is widely used in pharma to predict the concentration of a reactant over time. Half-life finds its applications in many fields of study such as pharmacokinetics, particle physics, carbon dating, etc.
- Blog
- Toshiba r830 bluetooth
- Dirt 4 reviews
- Free download hunting unlimited 2014
- Coins generator fifa 16 no survey
- Vsco image id
- Planet coaster requirements
- Pinnacle studio 15 versione prova
- Use hec ras
- Pokemon sun moon logo
- Microsoft toolkit 2-6-7 telecharger
- Tom jerry cartoon in urdu
- Apps like camerafi live
- Download muv luv steam 18- patch
- 1- imperialism 2- colony 3- social darwinism 4- civilize 5- superior 6- barbaric 7- empire
- Netgear n150 wireless usb adapter wna1100 version 1-5
- Persona 5 pc full free download mega
- Attack on titan anime soundtrack
- Ralink rt2870 mac driver
- I don t want to be a player no more reggae
- This way that mario party 2
- Carbon 14 half life
- Matrix path of neo pc free download
